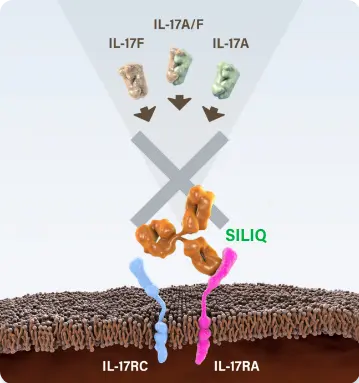
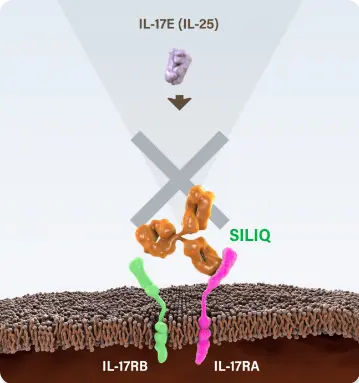
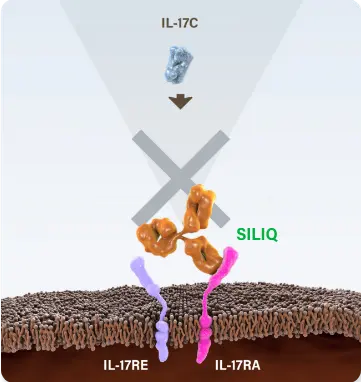
*SILIQ stops the action of a family of 4 proteins involved in psoriasis by blocking the gateway through which these proteins work. Other biologic medicines that work against this family of proteins target 1 protein from this group and have no effect on the other 3 proteins.1,8,9
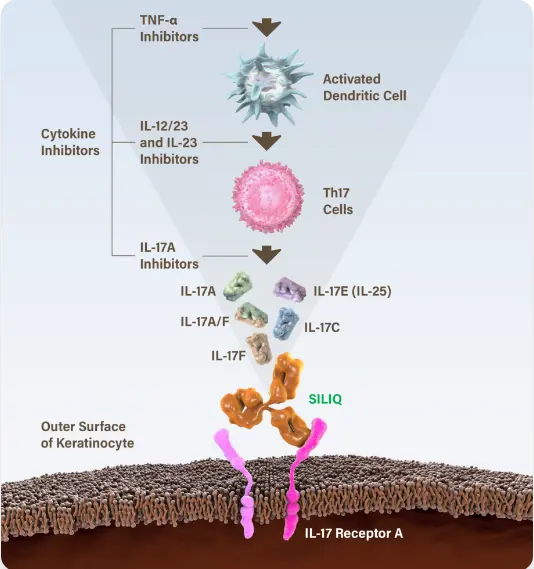
SILIQ blocks IL-17A, IL-17C, IL-17E (IL-25), IL-17A/F and IL-17F via their receptor to stop the proinflammatory cascade that leads to psoriasis.1
Blocking IL-17 Receptor A inhibits IL-17 cytokine-induced responses.1
The clinical significance of these characteristics is not fully known.

IL-17 cytokines are key players in the development of psoriasis.10-13
| Cytokine | In Psoriasis | Blocked by | Receptor Complex |
|---|---|---|---|
| IL-17A | Elevated 28x10 | SILIQ1 | IL-17RA/RC12 |
| IL-17C | Elevated 30x10 | SILIQ1 | IL-17RA/RE12 |
| IL-17E (IL-25) | Elevated11 | SILIQ1 | IL-17RA/RB13 |
| IL-17F | Elevated 33x10 | SILIQ1 | IL-17RA/RC12 |
| IL-17A/F | Elevated10 | SILIQ1 | IL-17RA/RC12 |
IL-17A inhibitors impede IL-17A.8,9 SILIQ blocks all of these IL-17 cytokines because it blocks interactions with the receptor that binds them: IL-17RA.1
A small study identifies the role of IL-17C in the development of psoriasis and characterizes it as the most abundant IL-17 cytokine in psoriatic lesional skin.14






It’s simple. Enrolling through the REMS portal takes just a few minutes.
INDICATION
SILIQ® injection is indicated for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy and have failed to respond or have lost response to other systemic therapies.
IMPORTANT SAFETY INFORMATION
WARNING: SUICIDAL IDEATION AND BEHAVIOR
Suicidal ideation and behavior, including completed suicides, have occurred in patients treated with SILIQ. Prior to prescribing SILIQ, weigh the potential risks and benefits in patients with a history of depression and/or suicidal ideation or behavior. Patients with new or worsening suicidal ideation and behavior should be referred to a mental health professional, as appropriate. Advise patients and caregivers to seek medical attention for manifestations of suicidal ideation or behavior, new onset or worsening depression, anxiety, or other mood changes [see Warnings and Precautions (5.1) in the full Prescribing Information].
Because of the observed suicidal behavior in subjects treated with SILIQ, SILIQ is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the SILIQ REMS Program [see Warnings and Precautions (5.2) in the full Prescribing Information].
Contraindications Do not use SILIQ in patients with Crohn's disease because SILIQ may cause worsening of disease; or in patients with clinically significant hypersensitivity to brodalumab or to any of the excipients in SILIQ or components of the container.
SILIQ Risk Evaluation and Mitigation Strategy (REMS) Program SILIQ is available only through a restricted program called the SILIQ REMS because of observed suicidal ideation and behavior in patients treated with SILIQ. Before prescribing SILIQ, prescribers must be certified with the program, have each patient sign a Patient-Prescriber Agreement Form, and provide the patient a Wallet Card describing symptoms requiring immediate medical evaluation. Pharmacies must be certified and only dispense to patients authorized to receive SILIQ. More information is available at SILIQREMS.com or by calling the SILIQ REMS Program Call Center at 855-511-6135855-511-6135.
Hypersensitivity Reactions Serious hypersensitivity reactions, including anaphylaxis requiring hospitalization, have been reported. If a serious hypersensitivity reaction occurs, immediately discontinue SILIQ and initiate appropriate therapy.
Infections SILIQ may increase the risk of infections. Serious infections and fungal infections were observed at a higher rate in patients treated with SILIQ than placebo-treated patients in clinical trials, including one case of cryptococcal meningitis that led to discontinuation of therapy.
Consider risks and benefits prior to prescribing SILIQ in patients with a chronic infection or history of recurrent infection.
Instruct patients to seek treatment if signs or symptoms of a chronic or acute infection occur.
Risk for Latent Tuberculosis (TB) Reactivation Evaluate patients for TB prior to initiating treatment with SILIQ and do not treat patients with active TB. Initiate treatment for latent TB prior to starting SILIQ and consider anti-TB therapy prior to initiation in patients with history of latent TB if adequate treatment cannot be confirmed. Monitor closely for symptoms of active TB during and after treatment.
Eczematous Eruptions Postmarketing cases of severe eczematous eruptions, including atopic dermatitis-like eruptions, have been reported. The onset of eczematous eruptions was variable, ranging from days to months after the first dose of SILIQ. Some cases of severe eczematous eruptions resulted in hospitalization. Treatment may need to be discontinued to resolve the eczematous eruption. Some patients with limited psoriasis treatment options were successfully treated for eczema while continuing SILIQ.
Crohn’s Disease In clinical trials, which excluded Crohn’s patients, one SILIQ patient was withdrawn after developing Crohn’s disease. Discontinue SILIQ if a patient develops Crohn’s disease.
Immunizations Avoid use of live vaccines in patients treated with SILIQ.
Adverse Reactions The most commonly reported adverse reactions in clinical trials were arthralgia, headache, fatigue, diarrhea, oropharyngeal pain, nausea, myalgia, injection site reactions, influenza, neutropenia, and tinea infections.
To report SUSPECTED ADVERSE REACTIONS, contact Bausch Health at 1-800-321-4576 or the FDA at 1-800-FDA-1088, or visit www.fda.gov/medwatch.
INDICATION
SILIQ® injection is indicated for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy and have failed to respond or have lost response to other systemic therapies.
Click here for full Prescribing Information, including Boxed Warning about suicidal ideation and behavior.
SILIQ® injection is indicated for the treatment of moderate to severe plaque psoriasis in adult patients who are candidates for systemic therapy or phototherapy and have failed to ...
WARNING: SUICIDAL IDEATION AND BEHAVIOR
Suicidal ideation and behavior, including completed suicides, have occurred in patients treated with SILIQ. Prior to prescribing SILIQ, weigh the potential risks and benefits in patients with a history of depression and/or ...
WARNING: SUICIDAL IDEATION AND BEHAVIOR
Suicidal ideation and behavior, including completed suicides, have occurred in patients treated with SILIQ. Prior to prescribing SILIQ, weigh the potential risks and benefits in patients with a history of depression and/or ...
